Fabrication of a chitosan‒Fe3O4/activated carbon/TiO\(_{2}\) nanocomposite as a Pb(II) heavy metal adsorbent
DOI:
https://doi.org/10.55713/jmmm.v36i2.2386Keywords:
CS-Fe3O4/AC/TiO2, adsorbent, heavy metal, superparamagnetism, industrial wasteAbstract
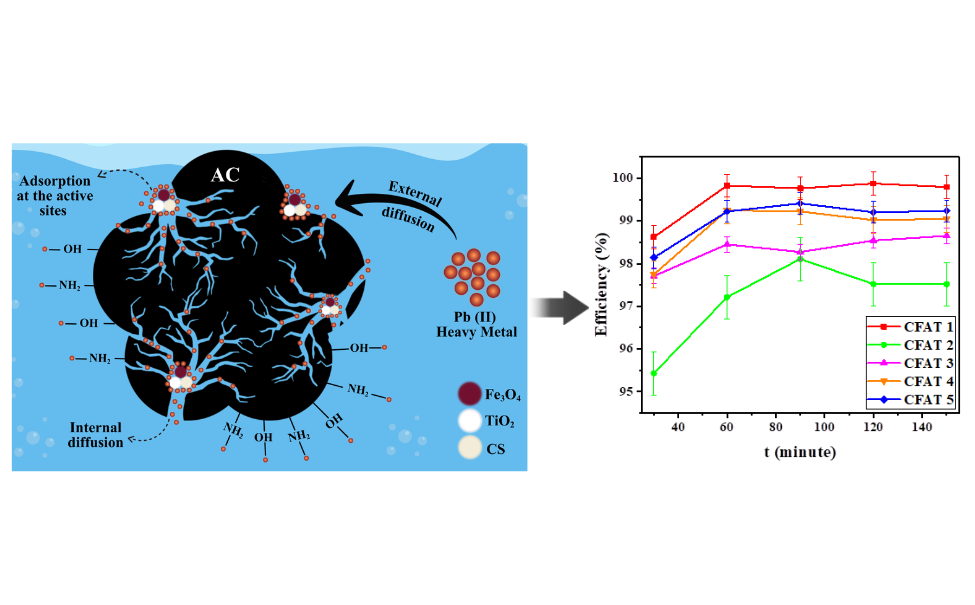
Nowadays, water pollution by heavy metal Pb(II) is a significant issue in many countries, including Indonesia. To overcome this problem, a suitable and efficient waste treatment method is necessary. Therefore, in this work, Fe3O4/AC/TiO2 nanocomposite modified with chitosan (CS) for the treatment of Pb (II) waste were investigated. CS was used due to its OH and NH2 functional groups, which enable the formation of a new hybrid nanocomposite that can be used repeatedly. To reduce production costs, environmentally friendly, raw natural materials, such as iron sand, coconut shells, and shrimp shells, were employed. The XRD characterization results indicate that the crystallite size of Fe3O4 is in the range of 16.05 nm to 24.52 nm, while that of TiO2 is 25.22 nm. The SEM‒EDX characterization indicates that the particle morphology is imperfectly round and aggregated. Furthermore, the FTIR analysis indicates the presence of N–H, Fe–O, C=O, and Ti–O–Ti functional groups, representing the CS, Fe3O4, AC, and TiO2 characteristics. The VSM results demonstrate that the CS-Fe3O4/AC/TiO2 exhibits superparamagnetic properties. In the Pb (II) heavy metal uptake test, CFAT 1 showed the most optimal results when the adsorption test was conducted for 120 min, resulting in an efficiency of 99.88%. Furthermore, this sample can be used repeatedly in four adsorption-desorption cycles. This suggests that the CS‒Fe3O4/AC/TiO2 nanocomposite can be an effective Pb(II) heavy metal absorbent.
Downloads
References
P. Higgins, R. Kumar, and S. H. Siddiqui, "Synthesis and characterization of SiO2/GO/Al2O3 nanocomposite adsorbent for the uptake of ciprofloxacin: Isothermal and reusability studies," Journal of the Iranian Chemical Society, vol. 22, no. 2, pp. 407–418, 2025. DOI: https://doi.org/10.1007/s13738-024-03158-9
M. S. Rostami, and M. M. Khodaei, "Recent advances in chitosan-based nanocomposites for adsorption and removal of heavy metal ions," International Journal of Biological Macromolecules, vol. 270, p. 132386, 2024. DOI: https://doi.org/10.1016/j.ijbiomac.2024.132386
Y. B. Nthwane, B. G. Fouda-Mbanga, M. Thwala, and K. Pillay, "A comprehensive review of heavy metals (Pb2+, Cd2+, Ni2+) removal from wastewater using low-cost adsorbents and possible revalorisation of spent adsorbents in blood fingerprint application," Environmental Technology, vol. 46, no. 3, pp. 414–430, 2025. DOI: https://doi.org/10.1080/09593330.2024.2358450
T. Shaikh, "Adsorption of Pb(II) from wastewater by natural and synthetic adsorbents," Biointerface Research in Applied Chemistry, vol. 10, no. 5, pp. 6522–6539, 2020. DOI: https://doi.org/10.33263/BRIAC105.65226539
S. Mohammadi, A. Kosari, H. Eslami, E. F. Moghadam, and A. Ghaffarian-Bahraman, "Toxic metal contamination in edible salts and its attributed human health risks: A systematic review and meta-analysis," Environmental Science and Pollution Research, vol. 32, no. 7, pp. 4313–4324, 2025. DOI: https://doi.org/10.1007/s11356-025-35940-4
B. Kayranli, O. Gök, T. Yilmaz, G. Gok, H. Çelebi, I. Y. Seçkin, and Ö. Ç. Mesutoğlu, "Low-cost organic adsorbent usage for removing Ni2+ and Pb2+ from aqueous solution and adsorption mechanisms," International Journal of Environmental Science and Technology, vol. 19, no. 5, pp. 3547–3564, 2022. DOI: https://doi.org/10.1007/s13762-021-03653-z
N. Abd-Talib, C. S. Chuong, S. H. Mohd-Setapar, U. A. Asli, K. F. Pa’ee, and K. Y. T. Len, "Trends in adsorption mechanisms of fruit peel adsorbents to remove wastewater pollutants (Cu(II), Cd(II) and Pb(II))," Journal of Water and Environment Technology, vol. 18, no. 5, pp. 290–313, 2020. DOI: https://doi.org/10.2965/jwet.20-004
K. Raj, and A. P. Das, "Lead pollution: Impact on environment and human health and approach for a sustainable solution," Environmental Chemistry and Ecotoxicology, vol. 5, pp. 79–85, 2023. DOI: https://doi.org/10.1016/j.enceco.2023.02.001
E. S. Soedjono, A. Slamet, N. Fitriani, M. S. Sumarlan, A. Supriyanto, D. R. M. Isnadina, and N. B. Othman, "Residual seawater from salt production (bittern) as a coagulant to remove lead (Pb2+) and turbidity from batik industry wastewater," Heliyon, vol. 7, no. 11, p. e08268, 2021. DOI: https://doi.org/10.1016/j.heliyon.2021.e08268
J. R. Miller, J. P. Gannon, and K. Corcoran, "Concentrations, mobility, and potential ecological risks of selected metals within compost amended, reclaimed coal mine soils, tropical South Sumatra, Indonesia," AIMS Environmental Science, vol. 6, no. 4, pp. 298–325, 2019. DOI: https://doi.org/10.3934/environsci.2019.4.298
L. Setyaningsih, F. Azhar Dikdayatama, and A. S. Wulandari, "Arbuscular mycorrhizal fungi and rhizobium enhance the growth of samanea saman (trembesi) planted on gold-mine tailings in Pongkor, West Java, Indonesia," Biodiversitas, Journal of Biological Diversity, vol. 21, no. 2, 2020. DOI: https://doi.org/10.13057/biodiv/d210224
G. K. Kinuthia, V. Ngure, D. Beti, R. Lugalia, A. Wangila, and L. Kamau, "Levels of heavy metals in wastewater and soil samples from open drainage channels in Nairobi, Kenya: Community health implication," Scientific Reports, vol. 10, no. 1, p. 8434, 2020. DOI: https://doi.org/10.1038/s41598-020-65359-5
A. A. Sabrin A, ‘Textile dye removal from wastewater effluents using chitosan-ZnO nanocomposite," Journal of Textile Science and Engineering, vol. 05, no. 03, pp. 5–8, 2015. DOI: https://doi.org/10.4172/2165-8064.1000200
H. Yousefzadeh, A. A. Salarian, and H. Sid Kalal, "Study of Pb(II) adsorption from aqueous solutions by TiO2 functionalized with hydroxide ethyl aniline (PHEA/n-TiO2)," Journal of Molecular Liquids, vol. 263, pp. 294–302, 2018. DOI: https://doi.org/10.1016/j.molliq.2018.03.023
Q. Guo, C. Zhou, Z. Ma, and X. Yang, "Fundamentals of TiO2 photocatalysis: Concepts, mechanisms, and challenges," Advanced Materials, vol. 31, no. 50, pp. 1–26, 2019. DOI: https://doi.org/10.1002/adma.201901997
J. F. Jeevakumari, and G. Suresh, "Comprehensive evaluation of solvothermally synthesized Fe3O4 and PEG capped Fe3O4 nanoparticles for magnetic and biomedical applications," Inorganic Chemistry Communications, vol. 174, p. 113904, 2025. DOI: https://doi.org/10.1016/j.inoche.2025.113904
K. Kalantari, M. B. Ahmad, H. R. F. Masoumi, K. Shameli, M. Basri, and R. Khandanlou, "Rapid adsorption of heavy metals by Fe3O4/talc nanocomposite and optimization study using response surface methodology," International Journal of Molecular, vol. 15, no. 7, pp. 12913–12927, 2014. DOI: https://doi.org/10.3390/ijms150712913
H. Zhan, Y. Bian, Q. Yuan, B. Ren, A. Hursthouse, and G. Zhu, "Preparation and potential applications of superparamagnetic Nano-Fe3O4," Processes, vol. 6, no. 4, pp. 1–22, 2018. DOI: https://doi.org/10.3390/pr6040033
D. Sartika, E. Malis, E. S. Mangunang, "Analisis distribusi nano-partikel Fe3O4 terhadap penyerapan logam berat Pb," Journal Teknologi Technoscientia, vol. 12 No.1, no. 1, pp. 8–11, 2019. DOI: https://doi.org/10.34151/technoscientia.v12i1.1777
S. Jafari, F. Zhao, D. Zhao, M. Lahtinen, A. Bhatnagar, and M. Sillanpää, "A comparative study for the removal of methylene blue dye by N and S modified TiO2 adsorbents," Journal of Molecular Liquids, vol. 207, pp. 90–98, 2015. DOI: https://doi.org/10.1016/j.molliq.2015.03.026
E. Asuquo, A. Martin, P. Nzerem, F. Siperstein, and X. Fan, "Adsorption of Cd(II) and Pb(II) ions from aqueous solutions using mesoporous activated carbon adsorbent: Equilibrium, kinetics and characterisation studies," Journal of Environmental Chemical Engineering, vol. 5, no. 1, pp. 679–698, 2017. DOI: https://doi.org/10.1016/j.jece.2016.12.043
B. W. Putri, A. Taufiq, C. I. Yogihati, S. T. U. I. Subadra, Sunaryono, N. Mufti, M. Diantoro, and M. S. A. Aziz, "Synthesis of nano-sized Mg0.2Fe2.8O4/AC/TiO2 composite for removal Pb(II) from water," AIP Conference Proceedings, vol. 3068, no. 1, p. 040005, 2024. DOI: https://doi.org/10.1063/5.0202502
I. Aranaz, A. Alcántara, M. C. Civera, C. Arias, B. Elorza, A. Heras, and F. N. A. Contreras, "Chitosan: An overview of its properties and applications," Polymers, vol. 13, no. 19, p. 3256, 2021. DOI: https://doi.org/10.3390/polym13193256
F. G. L. M. Borsagli, and A. Borsagli, "Chemically modified chitosan bio-sorbents for the competitive complexation of heavy metals ions: A potential model for the treatment of wastewaters and industrial spills," Journal of Polymers and the Environment, vol. 27, no. 7, pp. 1542–1556, 2019. DOI: https://doi.org/10.1007/s10924-019-01449-4
S. Begum, N. Y. Yuhana, N. Md Saleh, N. H. N. Kamarudin, and A. B. Sulong, "Review of chitosan composite as a heavy metal adsorbent: Material preparation and properties," Carbohydrate Polymers, vol. 259, p. 117613, 2021. DOI: https://doi.org/10.1016/j.carbpol.2021.117613
E. H. Sujiono, D. Zabrian, Zurnansyah, M.ulyati, V. Zharvan, Samnur, and N. A. Humairah, "Fabrication and characterization of coconut shell activated carbon using variation chemical activation for wastewater treatment application," Results in Chemistry, vol. 4, p. 100291, 2022. DOI: https://doi.org/10.1016/j.rechem.2022.100291
A. Taufiq, R. E. Saputro, Sunaryono, N. Hidayat, A. Hidayat, N. Mufti, M. Diantoro, A. Patriati, Mujamilah, E. G. R. Putra, and H. Nur, "Fabrication of magnetite nanoparticles dispersed in olive oil and their structural and magnetic investigations," IOP Conference Series Materials Science and Engineering, vol. 202, no. 1, p. 012008, 2017. DOI: https://doi.org/10.1088/1757-899X/202/1/012008
A. Taufiq, "Effects of the Annealing temperature on the structure evolution and antifungal performance of TiO2/Fe3O4 nanocomposites manufactured from natural sand," Nano, vol. 16, no. 2, pp. 1–12, 2021. DOI: https://doi.org/10.1142/S179329202150017X
K. M. Dimpe, J. C. Ngila, and P. N. Nomngongo, "Application of waste tyre-based activated carbon for the removal of heavy metals in wastewater," Cogent Engineering, vol. 4, no. 1, pp. 1–11, 2017. DOI: https://doi.org/10.1080/23311916.2017.1330912
E. Abu-Danso, S. Peräniemi, T. Leiviskä, and A. Bhatnagar, "Synthesis of s-ligand tethered cellulose nanofibers for efficient removal of Pb(II) and Cd(II) ions from synthetic and industrial wastewater," Environmental Pollution, vol. 242, no. part B, pp. 1988–1997, 2018. DOI: https://doi.org/10.1016/j.envpol.2018.07.044
A. Gholizadeh, "A comparative study of physical properties in Fe3O4 nanoparticles prepared by coprecipitation and citrate methods," Journal of the American Ceramic Society, vol. 100, no. 8, pp. 3577–3588, 2017. DOI: https://doi.org/10.1111/jace.14896
V. M. Vinosel, M. Asisi Janifer, S. Anand, and S. Pauline, "Structural and functional group characterization of nano-composite Fe3O4/TiO2 and its magnetic property," Mechanic, Materials Science & Engineering, vol 9, no. 1, pp. 01499407, 2017.
A. R. Liandi, A. H. Cahyana, R. T. Yunarti, and T. P. Wendari, "Facile synthesis of magnetic Fe3O4@chitosan nanocomposite as environmentally green catalyst in multicomponent Knoevenagel- Michael domino reaction," Ceramics International, vol. 48, no. 14, pp. 20266–20274, 2022. DOI: https://doi.org/10.1016/j.ceramint.2022.03.307
J. Ruey-Shin, Y. Yao-Chung, L. Chien-Shiun, L. Kuen-Song, L. His-Chuan, W. Sea-Fue, and S. An-Cheng, "Synthesis of magnetic Fe3O4/activated carbon nanocomposites with high surface area as recoverable adsorbents," Journal of the Taiwan Institute of Chemical Engineers, vol. 90, pp. 51–60, 2018. DOI: https://doi.org/10.1016/j.jtice.2017.12.005
S. Afzal, N. M. Julkapli, and L. K. Mun, "Visible light active TiO2/CS/Fe3O4 for nitrophenol degradation: Studying impact of TiO2, CS and Fe3O4 loading on the optical and photocatalytic performance of nanocomposite," Materials Science in Semi-conductor Processing, vol. 131, no. March, p. 105891, 2021. DOI: https://doi.org/10.1016/j.mssp.2021.105891
I. O. Wulandari, D. J. D. H. Santjojo, R. A. Shobirin, and A. Sabarudin, "Characteristics and magnetic properties of chitosan-coated Fe3O4 nanoparticles prepared by ex-situ co-precipitation method," RASĀYAN Journal of Chemistry, vol. 10, no. 4, pp. 1348–1358, 2017.
L. R. Herawati, A. Taufiq, H. Wisodo, N. Hidayat, and N. Mufti, "Antifungal activities of Fe3O4/Ag nanocomposites covered by diethylamine template," AIP Conference Proceedings,vol. 2748, p. 020035, 2023. DOI: https://doi.org/10.1063/5.0138545
A. Sheikhmohammadi, and J. Yeganeh, "Application of the Fe3O4-chitosan nano-adsorbent for the adsorption of metronidazole from wastewater : Optimization, kinetic, thermodynamic and equilibrium studies," vol. 164, pp. 694–706, 2020. DOI: https://doi.org/10.1016/j.ijbiomac.2020.07.188
N. Mufti, U. Munfarriha, A. Fuad, and M. Diantoro, "Synthesis and photocatalytic properties of Fe3O4@TiO2 core-shell for degradation of Rhodamine B," AIP Conference Proceedings, vol. 1712, no. 1, p. 050009, 2016. DOI: https://doi.org/10.1063/1.4941892
J. C. Ríos-Hurtado, E. M. Múzquiz-Ramos, A. Zugasti-Cruz, and D. A. Cortés-Hernández, "Mechanosynthesis as a simple method to obtain a magnetic composite (activated carbon/Fe3O4) for hyperthermia treatment," Journal of Biomaterials and Nano-biotechnology, vol. 07, no. 01, pp. 19–28, 2016. DOI: https://doi.org/10.4236/jbnb.2016.71003
S. N. Qoidah St. U. I. Subadra, A. Taufiq, N. Mufti, S. Sunaryono, N. Hidayat, E. Handoko, M. Alaydrus, and T. Amrillah, "Fe3O4/
MWCNT/TiO2 nanocomposites as excellent microwave absorber material," Journal of Alloys and Compounds, vol. 970, no. 1, p. 172590, 2024. DOI: https://doi.org/10.1016/j.jallcom.2023.172590
M. Liu, X. Zhang, Z. Li, L. Qu, and R. Han, "Fabrication of zirconium(IV)-loaded chitosan/Fe3O4/graphene oxide for efficient removal of alizarin red from aqueous solution," Carbohydrate Polymers, vol. 248, no. May, p. 116792, 2020. DOI: https://doi.org/10.1016/j.carbpol.2020.116792
J. K. Fatombi, "Adsorption of indigo carmine from aqueous solution by chitosan and chitosan/activated carbon composite: Kinetics, isotherms and thermodynamics studies," Fibers and Polymers, vol. 20, no. 9, pp. 1820–1832, 2019. DOI: https://doi.org/10.1007/s12221-019-1107-y
M. A. Shaker, and A. A. Yakout, "Optimization, isotherm, kinetic and thermodynamic studies of Pb(II) ions adsorption onto n-maleated chitosan-immobilized tio2 nanoparticles from aqueous media," Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, vol. 154, pp. 145–156, 2016. DOI: https://doi.org/10.1016/j.saa.2015.10.027
L. Qi, Z. Xu, X. Jiang, C. Hu, and X. Zou, "Preparation and antibacterial activity of chitosan nanoparticles," Carbohydrate Research, vol. 339, no. 16, pp. 2693–2700, 2004. DOI: https://doi.org/10.1016/j.carres.2004.09.007
H. Choi, "Applicability of composite beads, spent coffee grounds/ chitosan, for the adsorptive removal of Pb(II) from aqueous solutions," vol. 30, no. 5, pp. 536–545, 2019.
I. L. D. Wijayanti, and F. W. Mahatmanti, "Synthesis of chitosan/ activated carbon composite beads as an adsorbent of Pb(II) and Cu(II) ions in aqueous solution: A review," Indonesian Journal of Chemical Science, vol. 11, no. 2, pp. 190–197, 2022. DOI: https://doi.org/10.15294/ijcs.v11i2.54943
S. R. Mousavi-Qeydari, A. Samimi, D. Mohebbi-Kalhori, and E. Ahmadi, "A mesoporous melamine/chitosan/activated carbon biocomposite: Preparation, characterization and and its its application for Ni(II) uptake via ion imprinting," International Journal of Biological Macromolecules, vol. 188, no. August, pp. 126–136, 2021. DOI: https://doi.org/10.1016/j.ijbiomac.2021.08.020
N. Hanifah, St. U. I. Subadra, N. Hidayat, Sunaryono, C. I. Yogihati, W.A. Adi, Munasir, .T Amrillah, M. S. A. Aziz, and A. Taufiq, "A novel Fe3O4/ZnO/PANI/rGO nanohybrid material for radar wave absorbing," Materials Chemistry and Physics, vol. 317, p. 129169, 2024. DOI: https://doi.org/10.1016/j.matchemphys.2024.129169
J. S. Lee, J. M. Cha, H. Y. Yoon, J. K. Lee, and Y. K. Kim, "Magnetic multi-granule nanoclusters: A model system that exhibits universal size effect of magnetic coercivity," Scientific Reports, vol. 5, no. January, pp. 1–7, 2015. DOI: https://doi.org/10.1038/srep12135
L. Hou, Q. Liang, and F. Wang, "Mechanisms that control the adsorption–desorption behavior of phosphate on magnetite nanoparticles: The role of particle size and surface chemistry characteristics," RSC Advances, vol. 10, no. 4, pp. 2378–2388, 2020. DOI: https://doi.org/10.1039/C9RA08517C
J. Wang, and X. Guo, "Adsorption kinetic models: Physical meanings, applications, and solving methods," Journal of Hazardous Materials, vol. 390, p. 122156, 2020. DOI: https://doi.org/10.1016/j.jhazmat.2020.122156
Y. Raji, A. Nadi, M. Rouway, S. J. Sbai, W. Yassine, A. Elmahbouby, O. Cherkaoui, and S. Zyade, "Efficient adsorption of methyl orange on nanoporous carbon from agricultural wastes: Characterization, kinetics, thermodynamics, regeneration and adsorption mechanism," Journal of Composites Science, vol. 6, no. 12, p. 385, 2022. DOI: https://doi.org/10.3390/jcs6120385
H. Asadollahzadeh, M. Ghazizadeh, and M. Manzari, "Developing a magnetic nanocomposite adsorbent based on carbon quantum dots prepared from Pomegranate peel for the removal of Pb(II) and Cd(II) ions from aqueous solution," Analytical Methods in Environmental Chemistry Journal, vol. 4, no. 3, pp. 33–46, 2021. DOI: https://doi.org/10.24200/amecj.v4.i03.149
M. Keshvardoostchokami, L. Babaei, A. A. Zamani, A. H. Parizanganeh, and F. Piri, "Synthesized chitosan/iron oxide nanocomposite and shrimp shell in removal of nickel, cadmium and lead from aqueous solution," Global Journal of Environ-mental Science and Management, vol. 3, no. 3, pp. 267‒278, 2017.
M. I. Shariful, S. B. Sharif, J. J. L. Lee, U. Habiba, B. C. Ang, and M. A. Amalina, "Adsorption of divalent heavy metal ion by mesoporous-high surface area chitosan/poly (ethylene oxide) nanofibrous membrane," Carbohydrate Polymers, vol. 157, pp. 57–64, 2017. DOI: https://doi.org/10.1016/j.carbpol.2016.09.063
T. N. Dharmapriya, D. Li, Y.-C. Chung, and P.-J. Huang, "Green synthesis of reusable adsorbents for the removal of heavy metal ions," ACS Omega, vol. 6, no. 45, pp. 30478–30487, 2021. DOI: https://doi.org/10.1021/acsomega.1c03879
Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2026 Journal of Metals, Materials and Minerals

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish in this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial publication in this journal.












