Synthesis of zinc oxide photocatalysts from zinc-dust waste for organic dye degradation
DOI:
https://doi.org/10.55713/jmmm.v33i2.1608Keywords:
Zinc oxide, Zinc-dust waste, Hydrothermal, Photocatalyst, Dye degradationAbstract
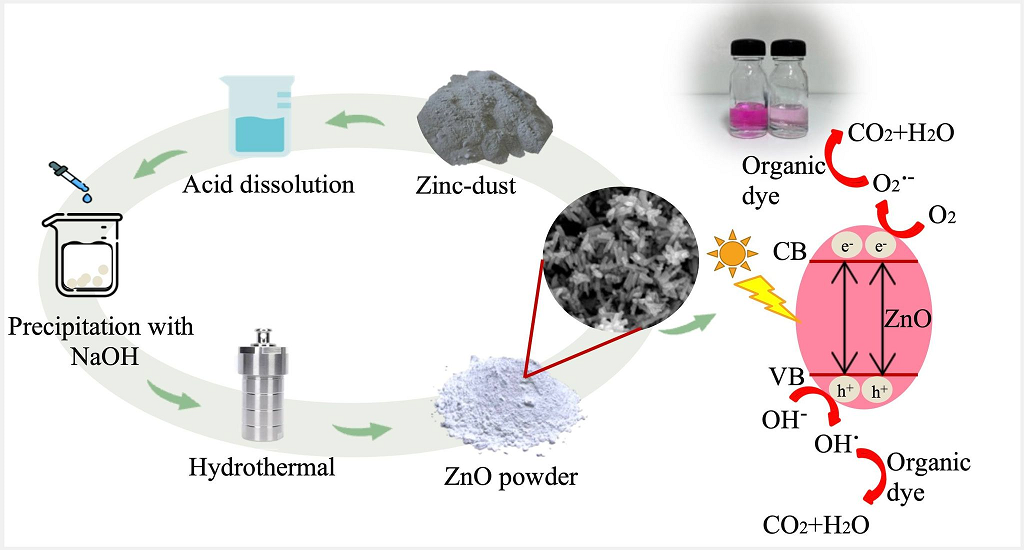
Zinc-dust waste from a hot-dip galvanizing plant in the metal plating industry was successfully used as the starting material to synthesize zinc oxide (ZnO) nanopowder via a hydrothermal method. Effects of acid types in zinc-dust dissolving process and concentration of NaOH for precipitation on physical characteristics and photocatalytic activity of the synthesized ZnO were investigated. Elemental composition, phase and crystallite size, morphology with chemical composition, and specific surface area of the ZnO nanostructures were characterized by X-ray fluorescence spectroscopy (XRF), X-ray diffraction (XRD), scanning electron microscopy with energy dispersive X-ray spectroscopy (SEM-EDS), and gas adsorption-desorption analysis, respectively. The photocatalytic performance of the prepared ZnO was evaluated by photodegradation of rhodamine B (RhB) under UV light irradiation. The ZnO nanostructures synthesized by dissolving zinc-dust with nitric acid, then precipitating with a solution of 6 M NaOH, and hydrothermal treatment at 170℃ for 8 h exhibited the highest dye degradation efficiency. It is up to 89.7% after UV irradiation for 240 min, which is comparable to the degradation efficiency of a commercial ZnO nanoparticles (92.7%). This work offers materials and synthesis method for alternative photocatalysts prepared from industrial waste that possess high photocatalytic activity for organic dye degradation.
Downloads
References
P. Sujaridworakun, and K. Natrchalayuth, "Influence of pH and HPC concentration on the synthesis of zinc oxide photocatalyst particle from zinc-dust waste by hydrothermal treatment," Advanced Powder Technology, vol. 25, pp. 1266-1272, 2014.
W. Liu, M. Wang, C. Xu, and S. Chen, "Facile synthesis of g-C3N4/ZnO composite with enhanced visible light photooxidation and photoreduction properties," Chemical Engineering Journal, vol. 209, pp. 386-393, 2012.
X. Tan, X. Wang, H. Hang, D. Zhang, N. Zhang, Z. Xiao, and H. Tao, "Self-assembly method assisted synthesis of g-C3N4/ZnO heterostructure nanocomposites with enhanced photocatalytic performance," Optical Materials, vol. 96, p. 109266, 2019.
G. Kamarajan, D. Benny Anburaj, V. Porkalai, A. Muthuvel, and G. Nedunchezhian, "Green synthesis of ZnO nanoparticles using Acalypha indica leaf extract and their photocatalyst degradation and antibacterial activity," Journal of the Indian Chemical Society, vol. 99, p. 100695, 2022.
O. Eskikaya, S. Ozdemir, G. Tollu, N. Dizge, R. Ramaraj, A. Manivannan, and D. Balakrishnan, "Synthesis of two different zinc oxide nanoflowers and comparison of antioxidant and photo- catalytic activity," Chemosphere, vol. 306, pp. 135389, 2022.
A. Ashar, I. A. Bhatti, T. Siddique, S. M. Ibrahim, S. Mirza, Z. A. Bhutta, M. Shoaib, M. Ali, M. B. Taj, M. Iqbal, S. Noor, and M. Mohsin, "Integrated hydrothermal assisted green synthesis of ZnO nano discs and their water purification efficiency together with antimicrobial activity," Journal of Materials Research and Technology, vol. 15, pp. 6901-6917, 2021.
N. S. Ridhuan, K. A. Razak, Z. Lockman, and A. A. Aziz, "Structural and morphology of ZnO nanorods synthesized using ZnO seeded growth hydrothermal method and its properties as UV sensing," PLOS ONE, vol. 15, p. 50405, 2012.
O. Lupan, and T. Pauporte, "Hydrothermal treatment for the marked structural and optical quality improvement of ZnO nanowire arrays deposited on light weight flexible substrates," Journal of Crystal Growth, vol. 312, pp. 2454-2458, 2010.
V. Gerbreders, M. Krasovska, E. Sledevskis, A. Gerbreders, I. Mihailova, E. Tamanis, and A. Ogurcovs, "Hydrothermal synthesis of ZnO nanostructures with controllable morphology change," Royal Society of Chemistry, vol. 22, pp. 1346-1358, 2022.
X. Chen, A. M. C. Ng, A. B. Djurisic, C. C. Ling, and W. K. Chan, "Hydrothermal treatment of ZnO nanostructures," Thin Solid Films, vol. 520, pp. 2656-2662, 2012.
P. Lad, V. Pathak, A. B. Thakkar, P. Thakor, M. P. Deshpande, and S. Pandya, “ZnO nanoparticles synthesized by precipitation method for solar-driven photodegradation of methylene blue dye and its potential as an anticancer agent,” Brazilian Journal of Physics, vol. 53, p. 63, 2023.
R. P. Cuapio, J. A. Alvarado, H. Juarez, and H-J. Sue, “Sun irradiated high efficient photocatalyst ZnO nanoparticles obtained by assisted microwave irradiation,” Materials Science and Engineering, vol. 289, p. 116263, 2023.
S. Sheikhi, M. Aliannezhadi, and F. S. Tehrani, “The effect of PEGylation on optical and structural properties of ZnO nanostructures for photocatalyst and photodynamic applications,” Materials Today, vol. 34, p. 105103, 2023.
S. N. Q. A. A. Aziz, K. C. Meng, S. Y. Pung, Z. Lockman, A. U. Hamid, and W. K. Tan, “Rapid growth of zinc oxide nanorods on kanthal wires by direct heating method and its photo-catalytic performance in pollutants removal,” Journal of Industrial and Engineering Chemistry, vol. 118, pp. 226-238, 2023.
T. M. Elmorsi, M. H. Elsayed, and M. F. Bakr, “Enhancing the removal of methylene blue by modified ZnO nanoparticles: kinetics and equilibrium studies,” Journal of Chemistry, vol. 95, pp. 590-600, 2017.
S. M. Ambalagi, M. Devendrappa, S. Nagaraja, and S. Sannakki, “Dielectric properties of PANI/CuO nanocomposites,” Materials Science and Engineering, vol. 310, p. 012081, 2018.
K. Zhao, B. Cao, J. Liu, Y. Wang, and L. An, “In-situ synthesis of Al76.8Fe24 complex metallic alloy phase in Al-based hybrid composite,” Journal of Materials Science & Technology, vol. 33, pp. 1177-1181, 2017.
A. Degen, and N. Kosec, “Effect of pH and impurities on the surface charge of zinc oxide in aqueous solution,” Journal of the European Ceramic Society, vol. 20, pp. 667-673, 2000.
K. Rupiyawet, K. Kaewlob, P. Sujaridworakun, P. Buggakupta, “Optimization of mixing conditions on the physical and tribological properties of brake pads” Key Engineering Materials, vol. 824, pp. 67-72, 2019.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Journal of Metals, Materials and Minerals

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish in this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial publication in this journal.












