Green acidic catalyst from cellulose extracted from sugarcane bagasse through pretreatment by electron beam irradiation and subsequent sulfonation for sugar production
DOI:
https://doi.org/10.55713/jmmm.v32i4.1540Keywords:
Cellulose, Acidic catalyst, Sulfonation, Pretreatment, Electron beam irradiation, Sugar productionAbstract
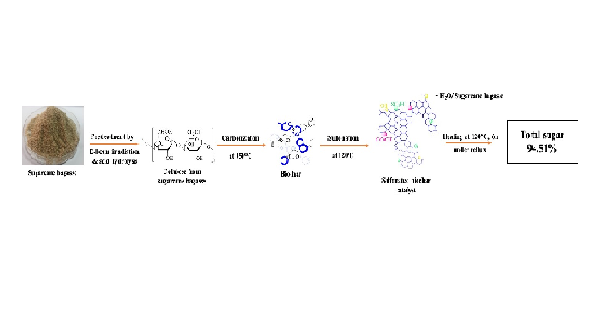
The objective of this research is to prepare a green acidic catalyst from cellulose derived from sugarcane bagasse (SB). Initially, SB was pretreated by electron beam irradiation with a dose of 50 kGy to 200 kGy and subsequent acid hydrolysis to obtain cellulose. The cellulose derived from SB was carbonized at different temperatures for 4 h and then sulfonated with heating at 120°C under reflux. Fourier transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) were used to confirm the successful preparation of acidic catalyst by irradiation pretreatment and subsequent sulfonation. The cellulose derived from irradiated SB at the lowest dose of 50 kGy was used as a representative irradiated sample for comparison with non-irradiation. Scanning electron microscope image of sulfonated biochar was observed pores with various sizes. The existence of sulfur atom onto sulfonated biochar surface was investigated by Energy dispersive X-ray spectroscopy. After sugar production by sulfonated biochar as an acidic catalyst, the total sugar content was measured by a phenol-sulfuric acid method using a UV-Vis spectrophotometer. The total sugar with 94.51 ± 1.35% content was found when the acidic catalyst was performed. It was remarkable to note that sulfonated biochar prepared from cellulose derived from SB after pretreatment and sulfonation exhibited outstanding result for being as an acidic catalyst for sugar production.
Downloads
References
N. Khumla, S. Sakuanrungsirikul, P. Punpee, T. Hamarn, T. Chaisan, L. Soulard, and P. Songsri, “Sugarcane breeding, germplasm development and supporting genetics research in thailand,” Sugar Tech, vol. 24, no. 1, pp. 193-209, 2022.
Y. Yang, H. Liu, M. Wu, J. Ma, and P. Lu, “Bio-based antimicrobial packaging from sugarcane bagasse nanocellulose/ nisin hybrid films,” International Journal of Biological Macromolecules, vol. 161, pp. 627-635, 2020.
D. E. C. Mazzeo, A. Misovic, F. A. Oliveira, C. E. Levy, J. Oehlmann, and M. R. R. de Marchi, “Effects of biostimulation by sugarcane bagasse and coffee grounds on sewage sludges, focusing agricultural use: Microbial characterization, respirometric assessment and toxicity reduction,” Waste Management, vol. 118, pp. 110-121, 2020.
M. de Melo Brites, A. A. Cerón, S. M. Costa, R. C. Oliveira, H. G. Ferraz, L. H. Catalani, and S. A. Costa, “Bromelain immobilization in cellulose triacetate nanofiber membranes from sugarcane bagasse by electrospinning technique,” Enzyme and Microbial Technology, vol. 132, pp. 109384, 2020.
S. N. H. M. Azmin, N. A. b. M. Hayat, and M. S. M. Nor, “Development and characterization of food packaging bioplastic film from cocoa pod husk cellulose incorporated with sugarcane bagasse fibre,” Journal of Bioresources and Bioproducts, vol. 5, no. 4, pp. 248-255, 2020.
M. T. Ban, N. Mahadin, and K. J. Abd Karim, “Synthesis of hydrogel from sugarcane bagasse extracted cellulose for swelling properties study,” Materials Today: Proceedings, vol. 50, pp. 2567-2575, 2022.
R. Wooley, M. Ruth, J. Sheehan, K. Ibsen, H. Majdeski, and A. Galvez, Lignocellulosic Biomass to Ethanol Process Design and Economics Utilizing Co-Current Dilute Acid Prehydrolysis and Enzymatic Hydrolysis Current and Futuristic Scenarios, National Renewable Energy Lab. (NREL), Golden, CO (United States), 1999.
P. Lanzafame, D. M. Temi, S. Perathoner, A. N. Spadaro, and G. Centi, “Direct conversion of cellulose to glucose and valuable intermediates in mild reaction conditions over solid acid catalysts,” Catalysis Today, vol. 179, no. 1, pp. 178-184, 2012.
R. A. Antonoplis, H. W. Blanch, R. P. Freitas, A. F. Sciamanna, and C. R. Wilke, “Production of sugars from wood using high-pressure hydrogen chloride,” Biotechnol Bioeng, vol. 25, no. 11, pp. 2757-2773, 1983.
K. Ohmine, H. Ooshima, and Y. Harano, “Kinetic study on enzymatic hydrolysis of cellulose by cellulose from Trichoderma viride,” Biotechnol Bioeng, vol. 25, no. 8, pp. 2041-2053, 1983.
B. Nidetzky, W. Steiner, M. Hayn, and M. Claeyssens, “Cellulose hydrolysis by the cellulases from Trichoderma reesei: a new model for synergistic interaction,” Biochem J, vol. 298, no. 3, pp. 705-710, 1994.
K.-E. Eriksson, “Enzyme mechanisms involved in cellulose hydrolysis by the rot fungus Sporotrichum pulverulentum,” Biotechnology and Bioengineering, vol. 20, no. 3, pp. 317-332, 1978.
G. W. Parshall, “Industrial applications of homogeneous catalysis: A review,” Journal of Molecular Catalysis, vol. 4, no. 4, pp. 243-270, 1978.
A. P. S. Chouhan, and A. K. Sarma, “Modern heterogeneous catalysts for biodiesel production: A comprehensive review,” Renewable and Sustainable Energy Reviews, vol. 15, no. 9, pp. 4378-4399, 2011.
S. Shanmugam, B. Viswanathan, and T. K. Varadarajan, “Esterification by solid acid catalysts—a comparison,” Journal of Molecular Catalysis A: Chemical, vol. 223, no. 1, pp. 143-147, 2004.
A. Yusuf, C. Snape, J. He, H. Xu, C. Liu, M. Zhao, G. Z. Chen, B. Tang, C. Wang, J. Wang, and S. N. Behera, “Advances on transition metal oxides catalysts for formaldehyde oxidation: A review,” Catalysis Reviews, vol. 59, no. 3, pp. 189-233, 2017.
W. Hölderich, M. Hesse, and F. Näumann, “Zeolites: Catalysts for organic syntheses,” Angewandte Chemie International Edition in English, vol. 27, no. 2, pp. 226-246, 1988.
S. B. Patil, P. R. Singh, M. P. Surpur, and S. D. Samant, “Cation‐exchanged resins: Efficient heterogeneous catalysts for facile synthesis of 1‐amidoalkyl‐2‐naphthols from one‐pot, three‐component condensations of amides/ureas, aldehydes, and 2‐naphthol,” Synthetic Communications, vol. 37, no. 10, pp. 1659-1664, 2007.
A. J. J Fripiat, and M. I. Cruz-Cumplido, “Clays as catalysts for natural processes,” Annual Review of Earth and Planetary Sciences, vol. 2, no. 1, pp. 239-256, 1974.
I. V. Kozhevnikov, “Advances in catalysis by heteropolyacids,” Russian Chemical Reviews, vol. 56, no. 9, pp. 811-825, 1987.
S. H. Y. S. Abdullah, N. H. M. Hanapi, A. Azid, R. Umar, H. Juahir, H. Khatoon, and A. Endut, “A review of biomass-derived heterogeneous catalyst for a sustainable biodiesel production,” Renewable and Sustainable Energy Reviews, vol. 70, pp. 1040-1051, 2017.
J. Bouchard, M. Méthot, and B. Jordan, “The effects of ionizing radiation on the cellulose of woodfree paper,” Cellulose, vol. 13, no. 5, pp. 601-610, 2006.
A. V. Ponomarev, and B. G. Ershov, “Radiation-induced high-temperature conversion of cellulose,” Molecules, vol. 19, no. 10, pp. 16877-16908, 2014.
E. Iller, A. Kukiełka, H. Stupińska, and W. Mikołajczyk, “Electron-beam stimulation of the reactivity of cellulose pulps for production of derivatives,” Radiation Physics and Chemistry, vol. 63, no. 3, pp. 253-257, 2002.
X. Fei, W. Jia, J. Wang, T. Chen, and Y. Ling, “Study on enzymatic hydrolysis efficiency and physicochemical properties of cellulose and lignocellulose after pretreatment with electron beam irradiation,” International Journal of Biological Macromolecules, vol. 145, pp. 733-739, 2020.
O. P. Sarosi, D. Bammer, E. Fitz, and A. Potthast, “Partial exchange of ozone by electron beam irradiation shows better viscosity control and less oxidation in cellulose upgrade scenarios,” Carbohydrate Polymers, vol. 265, pp. 118037, 2021.
M. L. E. Nagai, P. de Souza Santos, L. Otubo, M. J. A. Oliveira, and P. A. S. Vasquez, “Gamma and electron beam irradiation effects for conservation treatment of cellulose triacetate photographic and cinematographic films,” Radiation Physics and Chemistry, vol. 182, pp. 109395, 2021.
H. G. Kim, L. K. Kwac, Y.-S. Kim, H. K. Shin, and K.-Y. Rhee, “Synthesis and characterization of eco-friendly carboxymethyl cellulose based carbon foam using electron beam irradiation,” Composites Part B: Engineering, vol. 151, pp. 154-160, 2018.
S.-S. Wang, Y.-H. Han, Y.-X. Ye, X.-X. Shi, P. Xiang, D.-L. Chen, and M. Li, “Physicochemical characterization of high-quality bacterial cellulose produced by Komagataeibacter sp. strain W1 and identification of the associated genes in bacterial cellulose production,” RSC Advances, vol. 7, no. 71, pp. 45145-45155, 2017.
F. Lionetto, R. Del Sole, D. Cannoletta, G. Vasapollo, and A. Maffezzoli, “Monitoring wood degradation during weathering by cellulose crystallinity,” Materials, vol. 5, no. 10, pp. 1910-1922, 2012.
A. C. Lua, and T. Yang, “Effect of activation temperature on the textural and chemical properties of potassium hydroxide activated carbon prepared from pistachio-nut shell,” Journal of Colloid and Interface Science, vol. 274, no. 2, pp. 594-601, 2004.
G. Chen, and B. Fang, “Preparation of solid acid catalyst from glucose–starch mixture for biodiesel production,” Bioresource Technology, vol. 102, no. 3, pp. 2635-2640, 2011.
A. Mokhtari, M. Sabzi, and H. Azimi, “3D porous bioadsorbents based on chitosan/alginate/cellulose nanofibers as efficient and recyclable adsorbents of anionic dye,” Carbohydrate Polymers, vol. 265, pp. 118075, 2021.
M. Xia, W. Chen, J. Wu, Y. Chen, H. Yang, X. Chen, D. Zhu, and H. Chen, “Organic salt-assisted pyrolysis for preparation of porous carbon from cellulose, hemicellulose and lignin: New insight from structure evolution,” Fuel, vol. 291, pp. 120185, 2021.
Z. Zhang, M. Zhu, and D. Zhang, “A Thermogravimetric study of the characteristics of pyrolysis of cellulose isolated from selected biomass,” Applied Energy, vol. 220, pp. 87-93, 2018.
J. M. Fonseca, L. Spessato, A. L. Cazetta, K. C. Bedin, S. A. R. Melo, F. L. Souza, and V. C. Almeida, “Optimization of sulfonation process for the development of carbon-based catalyst from crambe meal via response surface methodology,” Energy Conversion and Management, vol. 217, pp. 112975, 2020.
A. d. C. Fraga, C. P. B. Quitete, V. L. Ximenes, E. F. Sousa-Aguiar, I. M. Fonseca, and A. M. B. Rego, “Biomass derived solid acids as effective hydrolysis catalysts,” Journal of Molecular Catalysis A: Chemical, vol. 422, pp. 248-257, 2016.
D. Zeng, Q. Zhang, S. Chen, S. Liu, and G. Wang, “Synthesis porous carbon-based solid acid from rice husk for esterification of fatty acids,” Microporous and Mesoporous Materials, vol. 219, pp. 54-58, 2016.
K. C. Bedin, A. L. Cazetta, I. P. A. F. Souza, O. Pezoti, L. S. Souza, P. S. C. Souza, J. T. C. Yokoyama, and V. C. Almeida, “Porosity enhancement of spherical activated carbon: Influence and optimization of hydrothermal synthesis conditions using response surface methodology,” Journal of Environmental Chemical Engineering, vol. 6, no. 1, pp. 991-999, 2018.
M. DuBois, K. A. Gilles, J. K. Hamilton, P. A. Rebers, and F. Smith, “Colorimetric method for determination of sugars and related substances,” Analytical Chemistry, vol. 28, no. 3, pp. 350-356, 1956.
C. Ban, S. Yang, H. Kim, and D. H. Kim, “Effect of Cu addition to carbon-supported Ru catalysts on hydrogenation of alginic acid into sugar alcohols,” Applied Catalysis A: General, vol. 578, pp. 98-104, 2019.
R. Zhong, F. Yu, W. Schutyser, Y. Liao, F. de Clippel, L. Peng, and B. F. Sels, “Acidic mesostructured silica-carbon nanocomposite catalysts for biofuels and chemicals synthesis from sugars in alcoholic solutions,” Applied Catalysis B: Environmental, vol. 206, pp. 74-88, 2017.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Journal of Metals, Materials and Minerals

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish in this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial publication in this journal.











