Morphology and electrochromic property of chemical bath deposited NiO films at different NiSO4 concentration
DOI:
https://doi.org/10.55713/jmmm.v32i4.1534Keywords:
NiO films, porous structure, electrochromic properties, chemical bath depositionAbstract
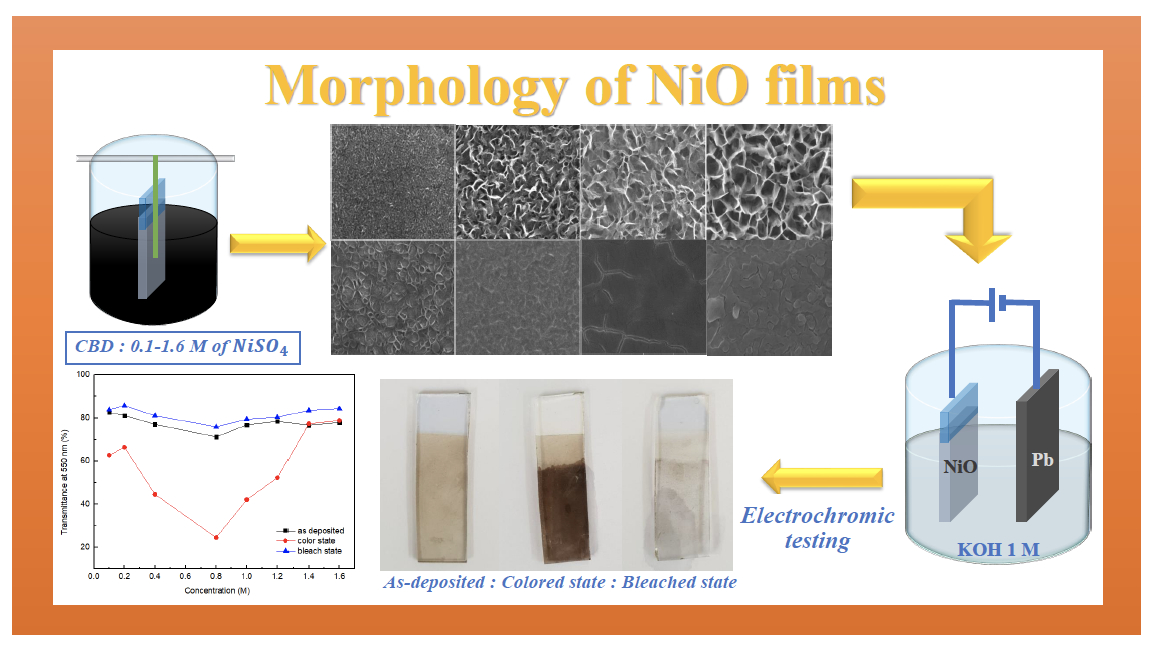
In this study, nickel oxide (NiO) films were prepared on indium tin oxide (ITO) glass by a chemical bath deposition (CBD) at different nickel sulfate (NiSO4) concentrations. The NiO films were verified for their structural properties with field emission scanning electron microscopy (FE-SEM) and X-ray diffraction (XRD) while the optical properties were investigated using a spectrophotometer. Moreover, the NiO films were studied to assess their electrochemical properties by cyclic voltammetry in potassium hydroxide (KOH) electrolyte. The results showed that annealed NiO films exhibited the dominant crystal structure of the (111) plane. Meanwhile, the NiSO4 concentration controlled morphological structure between dense and porous structures. The porous structure of NiO film was produced with the NiSO4 concentration in the range of 0.2 M to 1.0 M and the most porous structure was NiSO4 concentration at 0.8 M with a porosity of 64.56%. The optical contrast was calculated between bleached and colored states which were obtained at a maximum of 51.39% for NiO films at 0.8 NiSO4 concentration. Morphological effects and electrochromic properties were highly consistent. Analysis of the cyclic voltammetry (CV) results revealed that the cyclic stability for the highly porous structure of NiO films was more deteriorating than the less porous films.
Downloads
References
D. T. Gillaspie, R. C. Tenent, and A. C. Dillon, "Metal-oxide films for electrochromic applications: present technology and future directions," Journal of Materials Chemistry, vol. 20, pp. 9585-9592, 2010.
H. N. Ashtiani, A. Bahari, and S. Gholipour, "Investigation of coloration efficiency for tungsten oxide–silver nanocomposite thin films with different surface morphologies," Materials in Electronics, vol. 29, pp. 5820-5829, 2018.
C. Aiempanakit, A. Chanachai, N. Kanchai, M. Aiempanakit, and K. Aiempanakit, "Electrochromic property of tungsten trioxide films prepared by DC magnetron sputtering with oblique angle deposition and thermal oxidation," Journal of Metals, Materials and Minerals, vol. 31, no. 2, pp. 123-128, 2021.
C. Aiempanakit, R. Momkhunthod, and K. Aiempanakit, "Electro-chromism in nanoporous tungsten trioxide films prepared through anodization and thermal oxidation," Integrated Ferroelectrics, vol. 222, no. 1, pp. 84-92, 2022.
Q. Han, R. Wang, H. B. Zhu, M. X. Wan, and Y. H. Mai, "The preparation and investigation of all thin film electrochromic devices based on reactively sputtered MoO3 thin films," Materials Science in Semiconductor Processing, vol.126, pp.105686- 105695, 2021.
D. H. Choi, M. H Son, T. Y. Im, S.-H. Ahn, and C. S.-Y. Lee, "Microstructure control of NiO- based ion storage layer with various sized NiO particles to evaluate the electrochromic performance," Materials Chemistry and Physics, vol. 249, pp. 123121-123129, 2020.
S. Q. Jin, S. S. Wen, M. Y. Li, H. Zhong, Y. W. Chen, and H. G Wang, "Effect of the grain size on the electrochromic properties of NiO films," Optical Materials, vol. 109, pp.110280-110288, 2020.
D. R. Sahu, H. L. Yu, J. W. Tzu, C. W. Sheng, and L. H. Jow, " Synthesis and electrochromic property improvement of NiO films for device application," Thin Solid Films, vol. 707, pp. 138097-138107, 2020.
J. L. Hou, C. H. Wu, and S. J. Chang, " Electrochromic device integrated with GaInP/ GaAs/ Ge triple- junction solar cell," IEEE Electron. Device Lett, vol. 36, pp. 207-209, 2015.
X. Xia, Z. Ku, D. Zhou, Y. Zhong, Y. Zhang, Y. Wang, M. J. Huang, J. Tu, and H. J. Fan, "Perovskite solar cell powered electrochromic batteries for smart windows," Mater Horizons, vol. 3, pp. 588-595, 2016.
J. J. Shi, L. C. Lai, P. Zhang, H. L. Li, Y. M. Qin, Y. C. X. Gao, L. Luo, and J. Lu," Aluminum doped nickel oxide thin film with improved electrochromic performance from layered double hydroxides precursor in situ pyrolytic route," Journal of Solid State Chemistry, vol. 241, pp. 1-8, 2016.
S. Vijayakumar, S. Nagamuthu, and G. Muralidharan, "Super-capacitor studies on NiO nanoflakes synthesized through a microwave route," ACS Applied Materials & Interfaces, vol. 5, pp. 2188-2196, 2013.
W. J. Dong, Y. Lv, N. Zhang, L. L. Xiao, Y. Fan, and X. Y. Liu, "Trifunctional NiO–Ag–NiO electrodes for ITO-free electro-chromic supercapacitors," Journal of Materials Chemistry C, vol. 5, pp. 8408-8427, 2017.
A. C. Sonavane, A. I. Inamdar, P. S. Shinde, H. P. Deshmukh, R. S. Patil, and P.S. Patil, " Efficient electrochromic nickel oxide thin films by electrodeposition," Journal of Alloys and Compounds, vol. 489, pp. 667-673, 2010.
D. S. Dalavi, M. J. Suryavanshi, D. S. Patil, S. S. Mali, A. V. Moholkar, S. S. Kalagi,S. A. Vanalkar, S. R. Kang, J. H. Kim, and P. S. Patil, "Nanoporous nickel oxide thin films and its improved electrochromic performance: Effect of thickness," Applied Surface Science, vol. 257, pp. 2647-2656, 2011.
M. Martínez-Gil, M. I. Pintor-Monroy, M. Cota-Leal, D. Cabrera-German, A. Garzon- Fontecha, M. A. Quevedo- López, and M. Sotelo- Lerma, " Influence of annealing temperature on nickel oxide thin films grown by chemical bath deposition," Materials Science in Semiconductor Processing, vol. 72, pp. 37-45, 2017.
V. K. Patil, S. L. Pawar, M. N. Chougule, P. S. Godse, R. K. Sakhare, S. W. Sen, and P. D. Joshi, " Effect of annealing on structural, morphological, electrical and optical studies of nickel oxide thin films," Surface Engineered Materials and Advanced Technology, vol. 1, pp. 35-41, 2011.
Z. Bazhan, F. E. Ghodsi, and J. Mazloom, " Surface morphology, optical, and electrochromic properties of nanostructured nickel ferrite ( NiFe2O4) prepared by sol– gel method: effects of Ni/ Fe molar ratios," Applied Physics A, vol. 122, pp. 551-561, 2016.
S. A. Mahmoud, A. A. Akl, H. Kamal, and K. Abdel-Hady, "Opto-structural, electrical and electrochromic properties of crystalline nickel oxide thin films prepared by spray pyrolysis," Physica B, vol. 311, pp. 366-375, 2002.
R. Barir, B. Benhaou, S. Benhamida, A. Rahal, T. Sahraoui, and R. Gheriani, "Effect of precursor concentration on structural optical and electrical properties of NiO thin films prepared by spray pyrolysis," Journal of Nanomaterials, vol. 2017, pp. 10, 2017.
X. H. Xia, J. P. Tu, J. Zhang, X. L. Wang, W. K. Zhang, and H. Huang, "Morphology effect on the electrochromic and electrochemical performances of NiO thin films," Electrochimica Acta, vol. 53, pp. 5721-5724, 2008.
X. H. Xia, J. P. Tu, J. Zhang, X. L. Wang, W. K. Zhang, and H. Huang, "Electrochromic properties of porous NiO thin films prepared by a chemical bath deposition," Solar Energy Materials & Solar Cells, vol. 92, pp. 628-633, 2008.
J. R. Abenuz Acuña, I. Perez, V. Sosa, F. Gamboa, J. T. Elizalde, R. Farías, D. Carrillo, J. L. Enríquez, A. Burrola, and P. Mani, "Sputtering power effects on the electrochromic properties of NiO films," Optik - International Journal for Light and Electron Optics, vol. 231, pp. 166509-166524, 2021.
H. Liu, W. Zheng, X. Yan, and B. Feng, "Studies on electro-chromic properties of nickel oxide thin films prepared by reactive sputtering," Alloys and Compounds, vol. 462, pp. 356-361, 2008.
S.-Y. Han, D.-H. Lee, y.-j. chang, s.-o. ryu, t.-j. lee, and c.-h. chang, "the growth mechanism of nickel oxide thin films by room-temperature chemical bath deposition, " Journal of The Electrochemical Society, vol. 6, pp. 382-386, 2006
J. Wang, P. Yang, X. Wei, and Z. Zhou, "Preparation of NiO two-dimensional grainy films and their high- performance gas sensors for ammonia detection," Nanoscale Research Letters, vol. 10, pp. 1-6, 2015.
N. H. Al-Hardan, M. J. Abdullah, A. Abdul Aziz, H. Ahmad, and M. Rashid, "The effect of oxygen ratio on the crystallography and optical emission properties of reactive RF sputtered ZnO films," Physica B, vol. 405, pp. 1081-1085, 2010
R. Mechiakh, and R. BAensaha, "Analysis of Optical and Structural Properties of Sol–Gel TiO2 Thin Films," Moroccan Journal of Condensed Matter, vol. 7, pp. 54-57, 2006.
R. R. Reddy, and Y. Nazeer Ahamme, "A study on the Moss relation," Infrared. Phys. Technol, vol. 1, pp. 825-830, 1995
C. Salawan, M. Aiempanakit, K. Aiempanakit, C. Chananonnawathorn, P. Eiamchai, M. Horprathum, " Effects of oblique angle deposition on optical and morphological properties of WO3 nanorod films for electrochromic application," Materials Today: Proceedings, vol. 4, pp. 6423-6429, 2017.
J. Z. Ou, S. Balendhran, M. R. Field, D. G. McCulloch, A. S. Zoolfakar, R. A. Rani, S. Zhuiykov, A. P. O’Mullane and K. K.-zadeh, "The anodized crystalline WO3 nanoporous network with enhanced electrochromic properties," Nanoscale, vol. 4, pp. 5980-5988, 2012.
Y. Ren, W.K Chim, L. Guo, H. Tanoto, J. Pan, and S.Y Chiam, "The coloration and degradation mechanisms of electrochromic nickel oxide," Solar Energy Materials & Solar Cells, vol. 116, pp. 83–88, 2013.
[ 33] H. Yang, J. - H. Yu, H. J. Seo, R. H. Jeong, J. - H. Boo, " Improved electrochromic properties of nanoporous NiO film by NiO flake with thickness controlled by aluminum," Applied Surface Science, vol. 461, pp. 88-92, 2018.
R. Ismail, S. Ghafori, G. Kadhim, "Preparation and characterization of nanostructured nickel oxide thin films by spray pyrolysis," Applied Nanoscience, vol. 3, pp. 509-514, 2013.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2022 Journal of Metals, Materials and Minerals

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Authors who publish in this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial publication in this journal.












